Oncotarget published "Role for Fgr and Numb in retinoic acid-induced differentiation and G0 arrest of non-APL AML cells" which reported that retinoic acid is a fundamental regulator of cell cycle and cell differentiation.
Using a leukemic patient-derived in vitro model of a non-APL AML, these authors previously found that RA evokes activation of a macromolecular signaling complex, a signalosome, built of numerous MAPK-pathway-related signaling molecules; and this signaling enabled Retinoic-Acid-Response-Elements to regulate gene expression that results in cell differentiation/cell cycle arrest. Toward mechanistic insight into the nature of this novel signaling, they now find that the NUMB cell fate determinant protein is an apparent scaffold for the signalosome.
Numb exists in the cell bound to an ensemble of signalosome molecules, including Raf, Lyn, Slp-76, and Vav.
Fgr binds NUMB, which is associated with phosphorylation of NUMB and enhanced NUMB-binding and phosphorylation of select signalosome components, thereby betraying signalosome activation.
Signalosome activation is associated with cell differentiation along the myeloid lineage and G1/0 cell cycle arrest. If RA-induced Fgr expression is ablated by a CRISPR-KO; then the RA-induced phosphorylation of NUMB and enhanced NUMB-binding and phosphorylation of select signalosome components are lost. The cells now fail to undergo RA-induced differentiation or G1/0 arrest. In sum, this research team found that NUMB acts as a scaffold for a signaling machine that functions to propel RA-induced differentiation and G1/0 arrest, and that Fgr binding to NUMB turns the function on.
Dr. Andrew Yen from The Cornell University said, "Retinoic acid (RA) is a biologically fundamental regulator of cell proliferation and differentiation [1–3]. It is evolutionarily conserved and has widespread effects in numerous tissues."
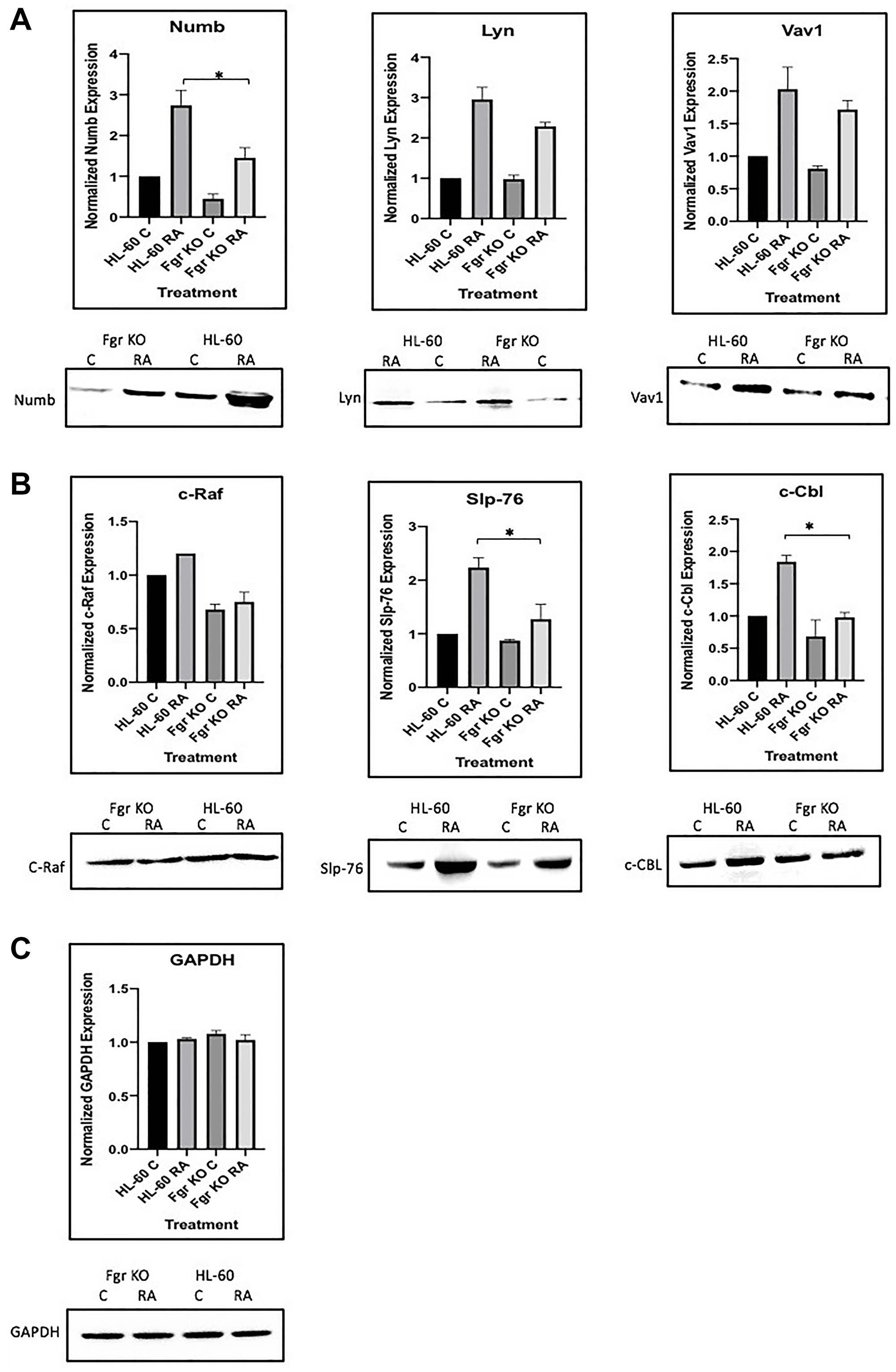
Figure 13: Regulation of “spine” molecules in HL-60 wt versus Fgr KO cells by RA. HL-60 wt and FGR KO cells were cultured for 72 h in the absence or presence of 1 μM RA as indicated and the whole cell lysate was collected. Twenty-five microgram of lysate per lane was run. (A) Western blots of SDS PAGE-resolved lysates were probed for Numb, Lyn and Vav 1 (n = 3). Films were scanned and bands of interest were quantified using ImageJ. Histograms show normalized densitometric values. Error bars indicate SEM. A representative blot, cropped to show only the band of interest, is included. (B) Western blots of c-Raf, Slp-76 and c-Cbl following the procedure described above. (C) Western blots of GAPDH were used as loading controls following the procedure described above. *p < .05 comparing RA-treated HL-60 wt samples to RA FGR KO cells samples.
It has been found that transcriptional activation by RAR/RXR to control cell differentiation and cell cycle arrest requires MAPK pathway related signaling to enable transcriptional activation necessary for driving cell differentiation/cell cycle G1/0 arrest.
The main finding is that the Numb fate determinant protein appears to act as a scaffold for the signalsome, binding a "spine" of molecules to which all other signalsome molecules are attached; and that RA induced expression of the Fgr SFK and its binding to Numb with phosphorylation of Numb, as well as other prominent signalsome phosphorylation events, indicating activation of the signalsome.
SLP-76 gets phosphorylated with RA-induced Fgr in parental wt, but not in Fgr KO, cells; so phosphorylation of Slp-76, one of the most prominent RA-induced p-tyr phosphorylations, where Slp-76 is known to IP with Fgr and Lyn, is also associated with Fgr binding Numb.
Numb also binds Lyn, which is known to become Y416 phosphorylated with RA treatment; indicating that Fgr binding Numb is associated with phosphorylation of Lyn, where Slp-76 and Lyn are the closest associates of Fgr in the signalsome force node representation of known IP partners.
In sum the Oncotarget data support a paradigm where signaling molecules bound to a Numb scaffold in a signalsome are activated by RA-induced Fgr expression and its binding to Numb to propel RA-induced differentiation and G1/0 arrest. In essence, RA is ergo increasing the amount of signalsome and increasing the amount of Fgr to activate them, thereby driving the cell to differentiate.
In sum the Oncotarget data support a paradigm where signaling molecules bound to a Numb scaffold in a signalsome are activated by RA-induced Fgr expression
The Yen Research Team concluded in their Oncotarget Research Output that a major challenge to RA differentiation therapy is the emergence of resistance to RA after the initial RA-treatment.
In an attempt to gain insight into how it develops, HL-60 cells resistant to RA were derived by sustained culture in progressively increasing RA concentrations.
A survey of expression of potentially responsible candidate molecules revealed that the biggest divergence between the parental cells and the RA-resistant derivatives was the failure of resistant cells to upregulate Fgr in response to RA.
Using drugs in lieu of Fgr to activate the signalsome in combination with RA may thus be a means of overcoming resistance.
Thus, the paradigm presented here may provide insights on candidates, the dysfunction of which are seminal to resistance.
DOI - https://doi.org/10.18632/oncotarget.27969
Full text - https://www.oncotarget.com/article/27969/text/
Correspondence to - Andrew Yen - ay13@cornell.edu
Keywords - retinoic acid(RA), FGR, Numb, non-APL AML, cell differentiation
About Oncotarget
Oncotarget is a bi-weekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit https://www.ImpactJournals.com or connect with @ImpactJrnls
Media Contact
MEDIA@IMPACTJOURNALS.COM
18009220957x105




